
The Eureka Crate from KiwiCo is for older children 14 years old and up. We have had a few of these stored away for a while, but last week, we finally opened up the Electrochemistry Lab and started working on it. These crates are a little more involved than the Kiwi (for ages 5-8) or Tinker Crates (for ages 9-14) and definitely require adult supervision. They also contain quite a few experiments that will likely take more than one day to do.

For the past week, we have been working on the Electrochemistry Lab crate. We have done two of the experiments so far and they’ve been very interesting! The second experiment even led to us looking through a microscope afterwards, just for fun!




It’s nice to have the crate materials list on the outside. My kids like looking at it since I don’t always let them open up the crates unless they will be doing them. I always appreciate how neatly everything is packed! Unfortunately, we aren’t able to get it repacked so neatly afterwards. The sturdy boxes also make good storage boxes, either for the crate materials for use again later or for storage in general.

All the crate materials laid out on the provided mess mat. The mess mat is a must!


Our first experiment was the aluminum eater, which was really interesting for the younger kids. It’s a dramatic reaction. The first picture above is a mistake. The copper sulfate ended up in the wrong cup, so we had to redo the measurements into a different cup, the second picture. Fortunately, there was enough copper sulfate to redo the measurements! One thing I do appreciate about our Kiwi Crates is that they always supply extra materials!

In this picture, Matthias is making the aluminum foil topper for one of the larger cups that came in the crate.. Make sure the foil forms a depression once you put it on the cup.



Make sure that the younger kids stay out of touching distance and that you or the older kids pour the teal mixture carefully onto the foil. The foil does get hot though we didn’t test out this fact ourselves! You can see that the teal mixture ate through the aluminum and left behind a residue.
The next experiment we did was not quite as exciting but it was still interesting to note the differences in voltage!

From left to right, we have 6 elements: magnesium, aluminum, copper, carbon, zinc, and iron (steel wool). Be careful with the steel wool, it flakes everywhere and you don’t want to get it in your eyes! None of us actually got it in our eyes, but I don’t think it would be a good thing.
We used these 6 elements to make a saltwater circuit.



We clipped the elements to the side of the cup then added water. Then we added salt (from a provided packet of sodium chloride) to the water. The salt dissolved in the water and formed the saltwater circuit.


In the first picture, the voltage reading is negative because the terminals are connected to the elements in reverse. To make the reading positive, we just had to switch the black terminal to where the red terminal was and the red terminal to where the black terminal was.

This is the completed experiment setup. The experiment book with the crate has a table to fill out so you can see the differences in voltages between pairs of connected elements.
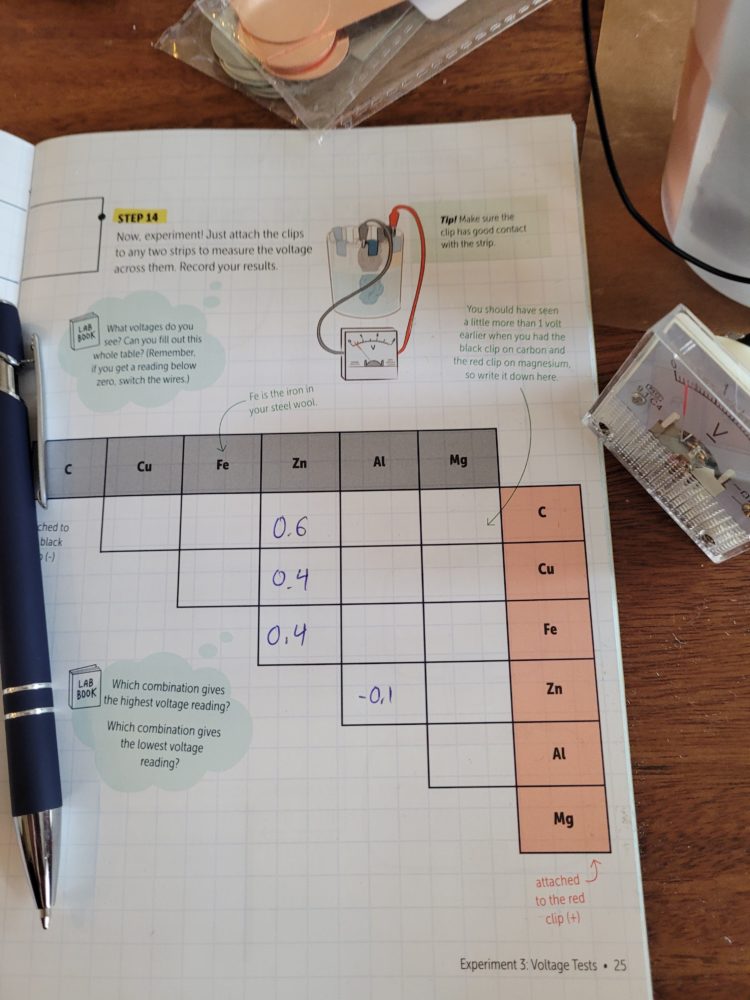
Here is our partially-filled table. This was interesting to fill out and then see which element combination provided the highest voltage reading! I won’t reveal it here so that if any of you do this experiment, you can be surprised!
We aren’t finished with the Electrochemistry Lab yet, but we will probably do at least another experiment in the next week. One experiment does require two cups to sit out overnight so that one will take some planning.
This particular Eureka crate is a little more expensive than the usual crates, but I think it was worth the cost with the 5 experiments you can do! My kids all had a good time either observing or doing the experiments. You will definitely want to have an adult present during the experiments though, just to make sure everyone stays back if needed or keeps materials where they should be.
If you’re interested in buying the Electrochemistry Lab, it is available in the KiwiCo store now by itself or as part of a 4-pack of Chemistry crates.
-Lynn


